New York, USA, March 02, 2026 (GLOBE NEWSWIRE) -- Acute Ischemic Stroke Market Poised for Expansion During the Forecast Period (2026–2036) as Innovative Treatment Modalities Gain Momentum | DelveInsight
The growth of the acute ischemic stroke market is expected to be driven primarily by increased awareness of symptoms, advances in acute stroke care, and a robust pipeline of potential candidates such as lenzocimab (Acticor Biotech), DM199 (DiaMedica Therapeutics), milvexian (BMS-986177) (Bristol Myers Squibb and Janssen Pharmaceuticals), BB-031 (Basking Biosciences) in the 7MM, according to DelveInsight.
DelveInsight’s Acute Ischemic Stroke Market Insights report includes a comprehensive understanding of current treatment practices, emerging acute ischemic stroke drugs, market share of individual therapies, and current and forecasted acute ischemic stroke market size from 2022 to 2036, segmented into leading markets (the US, EU4, UK, and Japan).
Acute Ischemic Stroke Market Summary
- The total acute ischemic stroke treatment market size is expected to grow positively by 2036 in the leading markets.
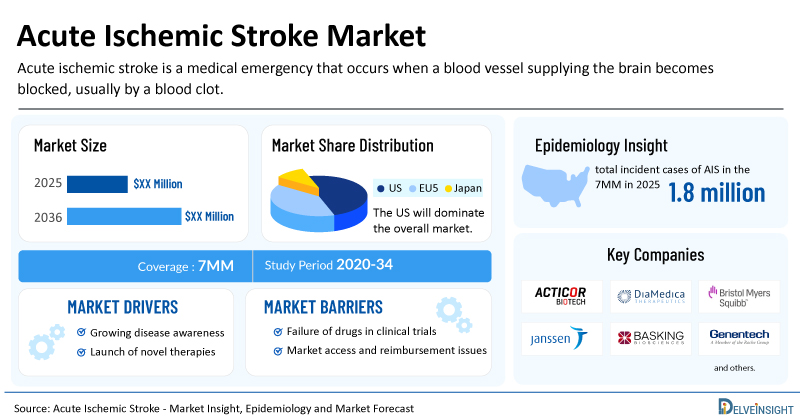
- The United States accounts for the largest market size of acute ischemic stroke, in comparison to EU4 (Germany, Italy, France, and Spain), the UK, and Japan.
- In 2025, the total incident cases of AIS were ~1.8 million in the 7MM.
- Key acute ischemic stroke companies, including Acticor Biotech, DiaMedica Therapeutics, Bristol Myers Squibb, Janssen Pharmaceuticals, Basking Biosciences, and others, are actively working on innovative acute ischemic stroke drugs.
- Some of the key acute ischemic stroke therapies in clinical trials include Glenzocimab, DM199, milvexian (BMS-986177), BB-031, LT3001, and others. These novel acute ischemic stroke therapies are anticipated to enter the acute ischemic stroke market in the forecast period and are expected to change the market.
Discover the acute ischemic stroke market size 2026–2036 @ https://www.delveinsight.com/sample-request/acute-ischemic-stroke-ais-market?utm_source=globenewswire&utm_medium=pressrelease&utm_campaign=spr
Ramandeep Singh, Senior Consultant of Forecasting and Analytics at DelveInsight, commented that a high degree of mortality and morbidity associated with ischemic stroke are two major factors likely to impede the AIS market success of the upcoming therapies.
Key Factors Driving the Growth of the Acute Ischemic Stroke Market
- Rising Prevalence of Stroke: The global burden of stroke, particularly ischemic stroke, continues to rise due to population growth and aging demographics. The increasing prevalence of stroke risk factors like hypertension, diabetes, obesity, and sedentary lifestyles is fueling case volumes.
- Novel Stem Cell & Thrombolytic Advances: Development of stem cell therapies and newer thrombolytic agents with a shorter half-life and a rapid onset of action might be safe and effective for AIS patients, giving pharma players wide opportunities
- Launch of Emerging AIS Drugs: The dynamics of the AIS market are anticipated to change in the coming years due to the launch of emerging drugs such as Glenzocimab (Acticor Biotech), DM199 (DiaMedica Therapeutics), milvexian (BMS-986177) (Bristol Myers Squibb and Janssen Pharmaceuticals), BB-031 (Basking Biosciences), LT3001 (Lumosa Therapeutics), and others.
Acute Ischemic Stroke Market Analysis
- Antiplatelet medication is the mainstay of care for treating stroke and transient ischemic episodes.
- Due to its affordability, accessibility, and relative safety, aspirin is the most extensively used medication.
- The US FDA has approved the gold-standard therapy, tissue plasminogen activator, r-tPA, often known as alteplase, to treat ischemic stroke.
- Further, in November 2020, the US FDA approved BRILINTA to reduce the risk of stroke in patients with an AIS.
- The evolving AIS clinical trial landscape, featuring candidates such as glenzocimab (Acticor Biotech), DM199 (DiaMedica Therapeutics), milvexian (BMS-986177) (Bristol Myers Squibb and Janssen Pharmaceuticals), BB-031 (Basking Biosciences), and others, is poised to drive growth in the AIS market.
Learn more about the acute ischemic stroke treatment landscape analysis @ Acute Ischemic Stroke Treatment Market
Acute Ischemic Stroke Competitive Landscape
Some of the AIS drugs in the clinical trials include Glenzocimab (Acticor Biotech), DM199 (DiaMedica Therapeutics), milvexian (BMS-986177) (Bristol Myers Squibb and Janssen Pharmaceuticals), BB-031 (Basking Biosciences), LT3001 (Lumosa Therapeutics), and others.
Acticor Biotech’s Glenzocimab (ACT017) is a humanized monoclonal antibody fragment that targets the platelet receptor GPVI, blocking thrombus formation while preserving normal hemostasis and thereby reducing bleeding risk, particularly intracranial bleeding. GPVI is found exclusively on platelets and megakaryocytes and binds to fibrin, fibrinogen, and collagen. In July 2022, the EMA granted glenzocimab PRIME status for stroke therapy.
The therapy has completed the Phase Ib/IIa ACTIMIS trial and the Phase II/III ACTISAVE trial as an adjunct to standard treatment for acute ischemic stroke (AIS). Results from ACTIMIS were presented at scientific meetings in 2022 and 2023 and published in Lancet Neurology in February 2024. Findings from ACTISAVE were disclosed at the 10th European Stroke Organization Conference (ESOC) in May 2024. The company is currently exploring strategic partnerships to further advance the development and commercialization of glenzocimab. The drug is also being studied in the Phase II/III GALICE trial for AIS.
DiaMedica Therapeutics’ DM199 (rinvecalinase alfa) is a recombinant form of human tissue kallikrein-1 (KLK1), a serine protease that influences blood flow, inflammation, fibrosis, oxidative stress, and neurogenesis. Its activity boosts the production of nitric oxide and prostaglandins. In February 2024, DiaMedica Therapeutics presented the design of the pivotal ReMEDy2 trial at the AHA International Stroke Conference, and the first patient was dosed in April 2024. An interim analysis is planned for Q2 2026.
Bristol Myers Squibb/Johnson and Johnson’s Milvexian (BMS-986177/JNJ-70033093) is an investigational, first-in-class, oral Factor XIa inhibitor intended to prevent and treat major thrombotic disorders. By blocking FXI, an enhancer of thrombin generation with only a modest role in normal hemostasis, milvexian may reduce vascular and thromboembolic risk while minimizing bleeding. It has received the US FDA Fast Track Designation for treating AIS.
Bristol-Myers Squibb and Janssen entered a global partnership in April 2018 to develop and commercialize BMS’s FXIa inhibitor portfolio, including BMS-986177. Milvexian is currently in the Phase III LIBREXIA-STROKE trial for preventing recurrent events after acute ischemic stroke or high-risk TIA, with registrational data expected in 2026. In May 2023, the FDA also granted Fast Track Designation for ischemic stroke, acute coronary syndrome, and atrial fibrillation within the broader LIBREXIA program.
The anticipated launch of these emerging therapies are poised to transform the acute ischemic stroke market landscape in the coming years. As these cutting-edge therapies continue to mature and gain regulatory approval, they are expected to reshape the acute ischemic stroke market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about leading companies in acute ischemic stroke market, visit @ Acute Ischemic Stroke Medication
Recent Developments in the Acute Ischemic Stroke Market
- In February 2026, Lumosa Therapeutics reported encouraging findings from two separate Phase 2 studies of its innovative stroke therapy, LT3001 (Odatroltide). LT3001 is a first-in-class treatment that provides both safe reperfusion and direct neuroprotection, aiming to overcome major shortcomings of existing acute ischemic stroke therapies.
- In November 2025, Basking Biosciences reported that it had begun dosing the first volunteers in its Phase 1 trial of BB-025, a fast-acting antidote engineered to selectively attach to and counteract BB-031, the company’s primary experimental treatment for acute ischemic stroke (AIS).
- In March 2025, the US FDA approved tenecteplase for the treatment of adults with acute ischemic stroke.
What is Acute Ischemic Stroke?
Acute ischemic stroke is a medical emergency that occurs when a blood vessel supplying the brain becomes blocked, usually by a blood clot. This blockage cuts off oxygen and nutrients to a specific part of the brain, causing brain cells to begin dying within minutes. Because these brain cells cannot regenerate, rapid treatment is critical to reduce long-term damage and improve the chances of recovery. Symptoms often appear suddenly and may include weakness on one side of the body, trouble speaking or understanding speech, vision problems, dizziness, or a severe headache. Early recognition and immediate medical attention are essential for the best possible outcome.
Acute Ischemic Stroke Epidemiology Segmentation
The acute ischemic stroke epidemiology section provides insights into the historical and current acute ischemic stroke patient pool and forecasted trends for the leading markets. In 2024, the US accounted for ~800,000 incident cases of AIS, which are expected to increase by 2034.
The acute ischemic stroke market report proffers epidemiological analysis for the study period 2022–2036 in the leading markets segmented into:
- Total Incident Cases of AIS
- Gender-specific Cases of AIS
- Age-specific Cases of AIS
- Type-specific Cases of AIS
Download the report to understand acute ischemic stroke incidence trends forecast @ Acute Ischemic Stroke Prevalence
| Acute Ischemic Stroke Market Report Metrics | Details |
| Study Period | 2022–2036 |
| Acute Ischemic Stroke Market Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Acute Ischemic Stroke Epidemiology Segmentation | Total Incident Cases of AIS, Gender-specific Cases of AIS, Age-specific Cases of AIS, and Type-specific Cases of AIS |
| Key Acute Ischemic Stroke Companies | Acticor Biotech, DiaMedica Therapeutics, Bristol Myers Squibb, Janssen Pharmaceuticals, Basking Biosciences, Genentech, Boehringer Ingelheim, AstraZeneca, and others |
| Key Acute Ischemic Stroke Therapies | Glenzocimab, DM199, milvexian (BMS-986177), BB-031, LT3001, TNKase/ Metalyse, Activase, BRILINTA, and others |
Scope of the Acute Ischemic Stroke Market Report
- Acute Ischemic Stroke Therapeutic Assessment: Acute Ischemic Stroke current marketed and emerging therapies
- Acute Ischemic Stroke Market Dynamics: Conjoint Analysis of Emerging Acute Ischemic Stroke Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Acute Ischemic Stroke Market Unmet Needs, KOL’s views, Analyst’s views, Acute Ischemic Stroke Market Access and Reimbursement
Discover more about acute ischemic stroke drugs in development @ Acute Ischemic Stroke Clinical Trials
Table of Contents
| 1 | Acute Ischemic Stroke Market Key Insights |
| 2 | Acute Ischemic Stroke Market Report Introduction |
| 3 | Acute Ischemic Stroke Market Overview at a Glance |
| 3.1 | Market Share (%) Distribution of AIS in 2025 |
| 3.2 | Market Share (%) Distribution of AIS in 2036 |
| 4 | Executive Summary |
| 5 | Key Events |
| 6 | Epidemiology and Patient Population |
| 7 | Disease Background and Overview |
| 7.1 | Introduction |
| 7.2 | Classification of Stroke |
| 7.3 | Clinical Manifestations |
| 7.4 | Etiology and Risk Factors |
| 7.5 | Pathophysiology of Ischemic Shock |
| 7.6 | Comorbidities Associated With AIS |
| 7.7 | Diagnosis of Ischemic Stroke |
| 7.8 | Management of Acute Ischemic Stroke |
| 7.9 | Clinical Guidelines |
| 8 | Epidemiology and Patient Population |
| 8.1 | Key Findings |
| 8.2 | Assumptions and Rationale |
| 8.3 | Total Incident Cases of AIS in the 7MM |
| 8.4 | The US |
| 8.4.1 | Incident Cases of AIS |
| 8.4.2 | Gender-specific Cases of AIS |
| 8.4.3 | Age-specific Cases of AIS |
| 8.4.4 | Type-specific Cases of AIS |
| 8.5 | EU4 and the UK |
| 8.6 | Japan |
| 9 | Acute Ischemic Stroke Patient Journey |
| 10 | Marketed Acute Ischemic Stroke Drugs |
| 10.1 | Key Cross Competition |
| 10.2 | TNKase/ Metalyse (tenecteplase): Genentech/ Boehringer Ingelheim |
| 10.2.1 | Product Description |
| 10.2.2 | Regulatory Milestone |
| 10.2.3 | Other Development Activities |
| 10.2.4 | Clinical Development |
| 10.2.5 | Safety and Efficacy |
| 10.3 | Activase (alteplase): Genentech/Boehringer Ingelheim |
| 10.4 | BRILINTA (ticagrelor): AstraZeneca |
| 11 | Emerging Acute Ischemic Stroke Therapies |
| 11.1 | Key Cross Competition |
| 11.2 | Glenzocimab (ACT017): Acticor Biotech |
| 11.2.1 | Drug Description |
| 11.2.2 | Other Development Activities |
| 11.2.3 | Clinical Development |
| 11.2.4 | Safety and Efficacy |
| 11.2.5 | Analysts' View |
| 11.3 | DM199 (rinvecalinase alfa): DiaMedica Therapeutics |
| 11.4 | Milvexian: Bristol Myers Squibb/Janssen Pharmaceutical |
| List to be continued in the final report.. | |
| 12 | AIS Market: Seven Major Market Analysis |
| 12.1 | Key Findings |
| 12.2 | Key Acute Ischemic Stroke Market Forecast Assumptions |
| 12.3 | Acute Ischemic Stroke Market Outlook |
| 12.4 | Attribute Analysis |
| 12.5 | Total Market Size of AIS in the 7MM |
| 12.6 | Total Market Size of AIS by Therapies in the 7MM |
| 12.7 | Market Size of AIS in the US |
| 12.7.1 | Total Market Size of AIS |
| 12.7.2 | The Market Size of AIS by Therapies |
| 12.8 | Market Size of AIS in EU4 and the UK |
| 12.9 | Market Size of AIS in Japan |
| 13 | Acute Ischemic Stroke Market SWOT Analysis |
| 14 | KOL Views on Acute Ischemic Stroke |
| 15 | Acute Ischemic Stroke Market Unmet Needs |
| 16 | Acute Ischemic Stroke Market Access and Reimbursement |
| 16.1 | The US |
| 16.2 | In EU4 and the UK |
| 16.3 | Japan |
| 16.4 | Market Access and Reimbursement of AIS |
| 17 | Bibliography |
| 18 | Acronyms and Abbreviations |
| 19 | Acute Ischemic Stroke Market Report Methodology |
Related Reports
Acute Ischemic Stroke Epidemiology
Acute Ischemic Stroke Epidemiology Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted AIS epidemiology in the 7MM, i.e., the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
Acute Ischemic Stroke Clinical Trial Analysis
Acute Ischemic Stroke Pipeline Insight – 2025 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key AIS companies including Tasly Pharmaceutical, Revalesio Corporation, Avilex Pharma, GNT Pharma, Biogen, AptaTargets, NC Medical Research, Prolong Pharmaceuticals, Saillant Therapeutics, TargED Biopharmaceuticals, and others.
Myocardial Infarction Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, market share of the individual therapies, and key myocardial infarction companies including AstraZeneca, Boehringer Ingelheim, Eli Lilly and Company, Amgen, Novartis, Idorsia Pharmaceuticals, Faraday Pharmaceuticals, CSL Behring, Immediate Therapeutics, Mitsubishi Chemical Group, Kancera, Bayer, Recardio, Mesoblast, and others.
Myocardial Infarction Clinical Trial Analysis
Myocardial Infarction Pipeline Insight – 2025 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key myocardial infarction companies including Novo Nordisk, CeleCor Therapeutics, Bayer, Shanghai Zhongshan Hospital, Takeda Pharmaceuticals, Nexel, Moleac Pte Ltd., LIB Therapeutics, Windtree Therapeutics, Faraday Pharmaceuticals, CSL Behring, Cardior Pharmaceuticals, AstraZeneca, R-Pharma, Shilpa Biologicals, and others.
Acute Myocardial Infarction Market
Acute Myocardial Infarction Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key AMI companies, including Idorsia Pharmaceuticals, Viatris, Recardio, AstraZeneca, Faraday Pharmaceuticals, CeleCor Therapeutics, Novo Nordisk, Bristol Myers Squibb, Johnson & Johnson Innovative Medicine, CellProthera, BioCardia, Kancera, Amgen, Arrowhead Pharmaceuticals, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.

Contact Us Shruti Thakur info@delveinsight.com +14699457679 www.delveinsight.com